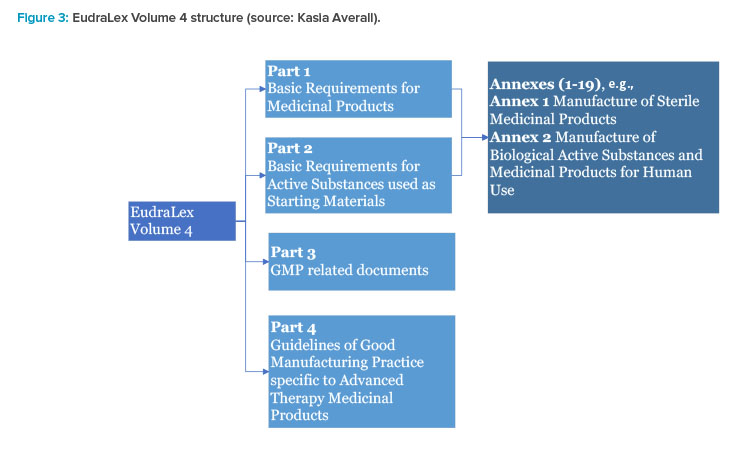
EUROPEAN COMMISSION Brussels, 03 February 2010 EudraLex The Rules Governing Medicinal Products in the European Union Volume 4 Go

EudraLex - Volume 10 Clinical trials guidelines and the impact of the new coming Regulation 536/2014

Dr.Prasadhi Shiva on LinkedIn: #quality #pharmaceutical #health #riskmanagement #fda #pharma #eugmp #qaqc

Details for: Good manufacturing practices (GMP) guidelines: the rules governing medicinal products in the European Union; EudraLex volume 4 concise references/ › TUS Midlands Library catalog

PPT - REVISION OF EUDRALEX VOL. 4 - GMP Luisa Stoppa, Ph.D. Inspection and Certification Department Italian Medicines Agency PowerPoint Presentation - ID:711040

PPT - REVISION OF EUDRALEX VOL. 4 - GMP Luisa Stoppa, Ph.D. Inspection and Certification Department Italian Medicines Agency PowerPoint Presentation - ID:711040

Amazon.com: The Handbook of Basic GMP Requirements: Collected guidelines from Eudralex Volume 4, Part I “Basic Requirements for Medicinal Products” (Italian Edition) eBook : Santoro, Karyn Noemi : Kindle Store